Efficiency of mechanical chest compressions with the LUCAS device in out-of-hospital cardiac arrest patients: a meta-analysis
Introduction
Cardiac arrest (CA) is the sudden cessation of cardiac ejection, severe ischemia and hypoxia of vital organs such as heart and brain, leading to the termination of life. This unexpected sudden death is also known as sudden death in medicine. CA is a combination of high incidence and low survival. About 350,000 out-of-hospital cardiac arrest (OHCA) cases occur in the United States each year (1-3). Although a series of new guidelines and standards were issued (1,2). The average survival rate for OHCA in the United States barely changed from 1978 to 2008 (4). With the development of science and technology and economy, people’s living standard is constantly improved, and the risk of cardiovascular disease is increased. Reducing the rate of OHCA is of great significance to the whole world.
Effective cardiopulmonary resuscitation (CPR) immediately after CA is of great significance to improve the prognosis and survival rate of patients, and is the key to avoid the occurrence of biological death (5,6). Chest compressions can raise the pressure inside the chest and press the heart directly to maintain blood flow, which plays a role in maintaining blood perfusion of important organs (6). There are many defects in manual chest compressions, such as the difference in CPR techniques caused by personnel training and the substandard CPR quality caused by personnel physical exertion, so that the inability to provide continuous chest compressions and other factors. Therefore, many scholars have studied various mechanical devices to replace manual CPR on the basis of the mechanism of cardiac compressions. The mechanism of chest compressions has been constantly updated and improved with the deepening of research. The use of mechanical chest compressions can avoid the fatigue of medical staff, and continue chest compressions at a consistent speed and depth, enabling medical staff to perform other critical tasks freely (7).
At present, the widely used mechanical chest compressors mainly include: (I) Automatic CPR system: Auto Pulse. It consists of an electric motor, a base plate, and a belt around the chest. Through the belt, the chest is compressed at a certain rate. (II) The Lund University Cardiac Assist System (LUCAS): LUCAS is a chest compression device that through the piston mode to perform chest compressions (7). It is usually powered by gas or batteries. A two-arm cylinder connected to a rigid back plate provides chest compressions and active decompression, after which the chest cage can fully spring back (5,7). However, previous studies have not determined the quality of artificial chest compressions and mechanical chest compressions. Therefore, this article focuses on the systematic review of previous studies to discuss the mechanical chest compressions efficiency of LUCAS device for patients with OHCA.
Methods
Retrieval strategy
Firstly, two authors (ML and KT) searched PubMed/Medline, EMBASE, Scopus, Cochrane Library, CNKI, Wanfang database from inception to February 20, 2019. Use the following keywords in English and Chinese joint corresponding retrieval: mechanical, manual, chest compression will, CA and CPR, LUCAS. The references of the literature were searched twice to reduce the omission.
Inclusion and exclusion criteria
The original study included in this study was a randomized controlled study. Subjects must include the LUCAS group and the manual group. The original article should compare the CPR effects of the two groups of OHCA. If subgroup data were to be included, the group with more cases was selected. Exclusion of cohort studies, case control studies, animal studies, meeting summaries, reviews, case reports, drug trials, literature not in English or Chinese, or in which full texts or incomplete data were not available.
Data extraction and quality evaluation
Two authors (ML and KT) read and extracted the literature information. Review Manager 5.3 bias score chart was used for quality evaluation. Inconsistencies in the process of data extraction and quality assessment shall be resolved through discussion.
Statistical analysis
The heterogeneity of each study was evaluated by Cochrane Q test and I2. When I2 ≥50%, there is heterogeneity between studies. To minimize bias, random effect models were selected for all meta-analysis steps in this study. The data in this study were counting data with odds ratio (OR) and confidence interval (CI) as the effect scale indicators. Meanwhile, funnel plot was drawn to evaluate publication bias. Except for Cochrane Q test, P<0.1 was considered statistically significant. All the others were defined as P<0.05, indicating a statistically significant difference. The RevMan5.3 software provided by the Cochrane collaboration was used for statistical analysis.
Results
Basic information of the included literature
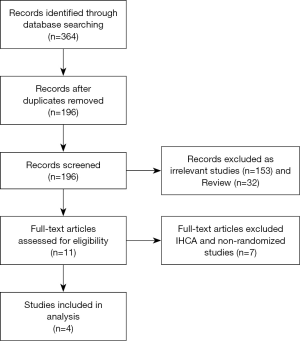
By searching the Chinese and English databases, a total of 364 original articles were obtained. After eliminating the repetition, reading the title and abstract of 365 articles, a total of 11 articles were basically satisfied with the research topic, including 2 non-randomized controlled trials (RCTs) and 5 articles for patients with CA in hospital, which were excluded and finally included in 4 articles (8-11). Baseline data are shown in Table 1. The literature retrieval process is shown in Figure 1.
Table 1
| Study | Study design | Study setting | Mechanical device | Population | Key outcomes | Support |
|---|---|---|---|---|---|---|
| Liu 2016 (11) | RCT | China | LUCAS | 71 OHCA patients | Survival to (ROSC) | No |
| Pekins 2015 (10) | RCT | USA | LUCAS | 4,471 OHCA patients | Survival to (ROSC/admission/30 days) | National institute for health research HTA-07-37-69 |
| Rubertsson 2014 (9) | RCT | Sweden | LUCAS | 2,589 OHCA patients | Survival to (ROSC/admission/discharge/30 days) | Institutional grants from Uppsala University and by Phsio-Control/Jolife AB |
| Smekal 2011 (8) | RCT | Sweden | LUCASTM | 148 OHCA patients | Survival to (ROSC/admission/discharge) | Uppsala University |
RCT, randomized controlled trial; LUCAS, Lund University Cardiac Assist System; OHCA, out-of-hospital cardiac arrest; ROSC, return of spontaneous circulation.
Return of spontaneous circulation (ROSC)
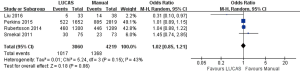
As shown in Figure 2, The success rate of ROSC in LUCAS group and Manual group was compared in all 4 articles (8-11). A total of 7,279 subjects were included. The success rate of ROSC in LUCAS group and Manual group is similar. The difference was not statistically significant [33.2% vs. 32.4%, OR =1.02, 95% CI: (0.85, 1.21), P=0.86].
Survival to hospital admission
As shown in Figure 3, A total of 3 articles (8-10) references and 7,208 subjects were included. There were 3,027 subjects in LUCAS group and 4,181 subjects in Manual group. There was no significant difference between the two groups in survival to hospital admission [23.2% vs. 23.4%, OR =0.99, 95% CI: (0.88, 1.10), P=0.80].
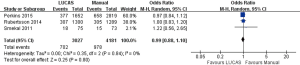
Survival to hospital discharge
As shown in Figure 4, a total of 2 articles (8,9) and 2,737 subjects were included. There were 1,375 subjects in LUCAS group and 1,362 subjects in Manual group. There was no significant difference between the two groups in survival to hospital discharge [8.3% vs. 7.9%, OR =1.06, 95% CI: (0.81, 1.40), P=0.68].

Survival to 30 days
As shown in Figure 5, a total of 2 articles (9,10) and 7,060 research subjects were included. There were 2,952 subjects in LUCAS group and 4,108 subjects in Manual group. Survival to 30 days was not significantly different between the two groups [7.1% vs. 7.0%, OR =1.00, 95% CI: (0.82, 1.21), P=0.96].
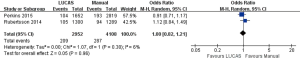
Quality evaluation and publication bias
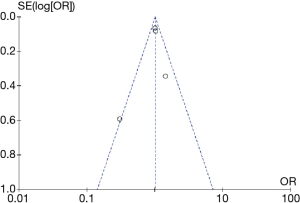
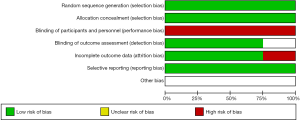
In this study, the bias analysis tool Review Manager 5.3 was used to grade each article, due to the need of patient rescue, double-blind design cannot be carried out, Therefore, the blind method score of each study is low, the score of other indicators is high, and the sample size of the study is large, so the overall quality is high (Figure S1). Results as shown in Figure 6, the included articles were distributed on both sides of the central line, which was relatively symmetrical and located at the tip of the funnel, indicating that the articles were of high quality and the publication deviation was small.
Discussion
High-quality CPR is of great significance to improve the prognosis and survival rate of CA (6). The 2015 American heart association guidelines and the 2015 European resuscitation commission guidelines both emphasize the importance of CPR. It is emphasized that high-quality CPR should be provided with sufficient frequency, chest compression depth, minimum compression interval and avoid excessive ventilation (1,6). The advantage of manual CPR as a traditional CPR method is that it can quickly intervene in the rescue of patients, but the quality of CPR will be seriously reduced over time. Since the machine will not feel fatigue, can ensure enough frequency, chest compression depth, minimum compression gap (12). The LUCAS is a chest compression device that through the piston mode to perform chest compression (12). It provides chest compressions and active decompression. The chest cage can completely rebound after pressure (5,7).
Previous studies have not concluded the comparison between LUCAS chest compressive efficiency and manual chest compressive efficiency. Some studies have shown that mechanical chest compressions are effective (12-15). It can improve the prognosis of patients and ROSC rate. Some studies also suggested no difference between LUCAS and manual chest compressive (16). Some studies suggest that manual chest compressions are better (17,18). The mechanical chest compressive device was less capable of providing ROSC than the manual group, and the 30-day survival rate and neurological outcome of the manual group were better than those of the mechanical group. This is probably because: firstly, the first defibrillation in the mechanical group may be 1.5 minutes later than in the manual group, and the installation of the device may interrupt the continuity of the press. Secondly, some patients may be unable to use the device because of their size, in the LINK trial (5,7), we know that only 5% of patients are expected to be eligible for LUCAS. Finally, it may be related to the occurrence of device-related adverse events, such as tension pneumothorax and visceral damage caused by the application of mechanical equipment. Khan et al. found that the relative risk of tension pneumothorax induced by compression in the manual group was 44% lower than that in the LUCAS group [OR =0.56, 95% CI: (0.08, 3.38)]. The probability analysis also showed that the manual group was the most secure (SUCAR, 71%) (5,7). Compared with AutoPulse, both the manual group [OR =0.15, 95% CI: (0.01, 0.73)] and the LUCAS group [OR =0.07, 95% CI: (0.00, 0.43)] reduced the risk of hematoma formation (5,7). However, this does not mean that all complications were low in the manual group. In terms of hemothorax and hematomas, probability analysis showed that compared with LUCAS, AutoPulse and manual group, LUCAS had the highest safety, hemothorax (SUCAR, 88%), and hematothorax (SUCAR, 94%). AutoPulse performed best in terms of visceral damage (SUCAR, 56%). Therefore, this article focuses on the systematic review of previous studies, and discusses the advantages and disadvantages of LUCAS group and manual group for patients with OHCA from four aspectS
Finally, four groups of RCTs were included in this paper, with a total of 7,279 subjectS The efficiency of the two groups was analyzed and compared from four aspects: ROSC, survival to hospital admission, survival to hospital discharge, and survival to hospital admission. The results showed that there was no difference in the four aspectS
Study limitations
The reasons for the large heterogeneity of the results of this study may be as follows: Firstly, the Review Manager 5.3 bias analysis tool was used to score each literature in this study. Due to the need of rescuing patients, the double-blind design could not be carried out, so the blind method score of each study was relatively low. Secondly, there are differences in equipment type, equipment use time, artificial CPR quality, prognosis and nursing quality, which can lead to significant differences. Thirdly, significant clinical heterogeneity exists between the study samples, and our analysis results may be questioned. Fourthly, there is insufficient research on the recovery of neurological function in prognosis, but it is generally considered as an important result by clinicians and patientS Finally, the study included two large multicenter studies and two relatively small sample trials. This leads to potential bias against the main findings.
Conclusions
the meta-analysis showed there is no difference in the efficacy of the LUCAS device in OHCA compared to the manual group. These may be due to: firstly, the mechanical device wasted the best time in the process of installation and could not be used for CPR immediately as the manual group. Secondly, the mechanical device is not suitable for specific patientS Finally, the mechanical device may more easily lead to the formation of pneumothorax or hematoma. Mechanical chest compressions cannot replace manual chest compressions, but they can be used as adjuvant therapeutic devices to liberate medical staff and avoid fatigue. It is able to continue chest compressions at a consistent rate and depth, allowing medical personnel the freedom to perform other critical tasks. The efficacy of the LUCAS device in OHCA requires a large number of high-quality studies, especially well-documented randomized trials.
Acknowledgments
Funding: The study was funded by Scientific Research Development Program of North Sichuan Medical College (CBY16-A-ZD10) and Nanchong Government-University Strategic Cooperation Project in Science and Technology (18SXHZ0505).
Footnote
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at http://dx.doi.org/10.21037/jxym.2020.02.02). The authors have no conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Kleinman ME, Brennan EE, Goldberger ZD, et al. Part 5: Adult Basic Life Support and Cardiopulmonary Resuscitation Quality: 2015 American Heart Association Guidelines Update for Cardiopulmonary Resuscitation and Emergency Cardiovascular Care. Circulation 2015;132:S414-35. [Crossref] [PubMed]
- Perkins GD, Handley AJ, Koster RW, et al. European Resuscitation Council Guidelines for Resuscitation 2015: Section 2. Adult basic life support and automated external defibrillation. Resuscitation 2015;95:81-99. [Crossref] [PubMed]
- Riess ML. New developments in cardiac arrest management. Adv Anesth 2016;34:29-46. [Crossref] [PubMed]
- Ewy GA. Cardiocerebral and cardiopulmonary resuscitation - 2017 update. Acute Med Surg 2017;4:227-34. [Crossref] [PubMed]
- Perkins GD, Brace S, Gates S. Mechanical chest-compression devices: current and future roles. Curr Opin Crit Care 2010;16:203-10. [Crossref] [PubMed]
- Idris AH, Guffey D, Pepe PE, et al. Chest compression rates and survival following out-of-hospital cardiac arrest. Crit Care Med 2015;43:840-8. [Crossref] [PubMed]
- Couper K, Smyth M, Perkins GD. Mechanical devices for chest compression: to use or not to use? Curr Opin Crit Care 2015;21:188-94. [Crossref] [PubMed]
- Smekal D, Johansson J, Huzevka T, et al. A pilot study of mechanical chest compressions with the LUCAS™ device in cardiopulmonary resuscitation. Resuscitation 2011;82:702-6. [Crossref] [PubMed]
- Rubertsson S, Lindgren E, Smekal D, et al. Mechanical chest compressions and simultaneous defibrillation vs conventional cardiopulmonary resuscitation in out-of-hospital cardiac arrest: the LINC randomized trial. JAMA 2014;311:53-61. [Crossref] [PubMed]
- Perkins GD, Lall R, Quinn T, et al. Mechanical versus manual chest compression for out-of-hospital cardiac arrest (PARAMEDIC): a pragmatic, cluster randomised controlled trial. Lancet 2015;385:947-55. [Crossref] [PubMed]
- Liu Y. Comparison of the effects of unarmed and mechanical external chest compression in out-of-hospital cardiac arrest patientS Chin J Emerg Resusc Disaster Med 2016;10:657-9.
- Westfall M, Krantz S, Mullin C, et al. Mechanical versus manual chest compressions in out-of-hospital cardiac arrest: a meta-analysis. Crit Care Med 2013;41:1782-9. [Crossref] [PubMed]
- Anantharaman V, Ng BL, Ang SH, et al. Prompt use of mechanical cardiopulmonary resuscitation in out-of-hospital cardiac arrest: the MECCA study report. Singapore Med J 2017;58:424-31. [Crossref] [PubMed]
- Halperin HR, Paradis N, Ornato JP, et al. Cardiopulmonary resuscitation with a novel chest compression device in a porcine model of cardiac arrest: improved hemodynamics and mechanisms. J Am Coll Cardiol 2004;44:2214-20. [Crossref] [PubMed]
- Couper K, Yeung J, Nicholson T, et al. Mechanical chest compression devices at in-hospital cardiac arrest: a systematic review and meta-analysis. Resuscitation 2016;103:24-31. [Crossref] [PubMed]
- Ji C, Lall R, Quinn T, et al. Post-admission outcomes of participants in the PARAMEDIC trial: a cluster randomised trial of mechanical or manual chest compressions. Resuscitation 2017;118:82-8. [Crossref] [PubMed]
- Khan SU, Lone AN, Talluri S, et al. Efficacy and safety of mechanical versus manual compression in cardiac arrest - a Bayesian network meta-analysis. Resuscitation 2018;130:182-8. [Crossref] [PubMed]
- Li H, Wang D, Yu Y, et al. Mechanical versus manual chest compressions for cardiac arrest: a systematic review and meta-analysis. Scand J Trauma Resusc Emerg Med 2016;24:10. [Crossref] [PubMed]
Cite this article as: Liu M, Tang K, Ai J, Shuai Z, Zheng J, Gou J, Lv Z. Efficiency of mechanical chest compressions with the LUCAS device in out-of-hospital cardiac arrest patients: a meta-analysis. J Xiangya Med 2020;5:3.